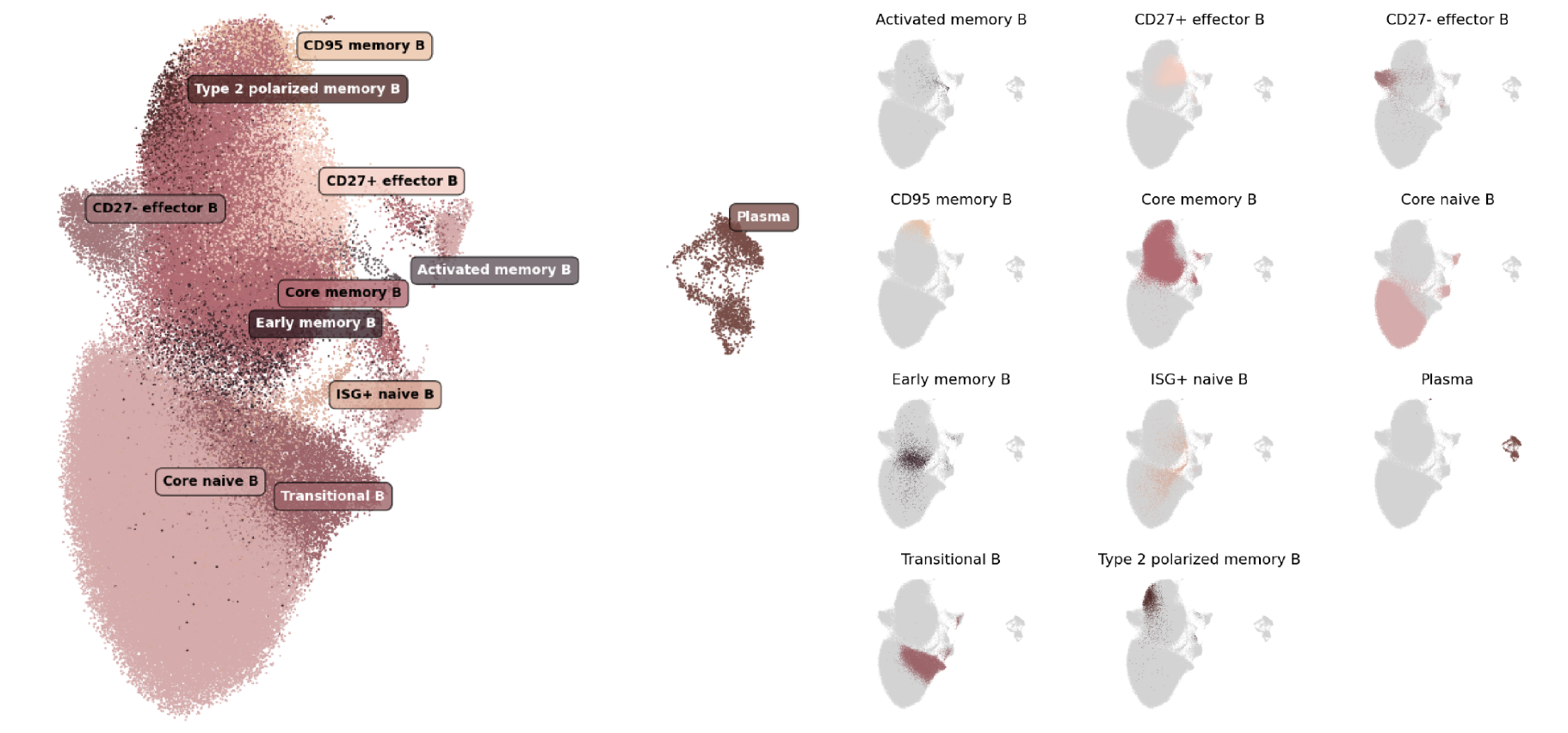
In our cell type atlas, we define B cell and Plasma cell types at 3 levels of resolution, from one Level 1 type (broadest) to 11 Level 3 types (highest resolution) based on marker gene expression and cell types previously described in literature. Descriptions of the marker expression criteria are provided below.
The hierarchical relationships between types, the usage of labels at each level of our cell type hierarchy, and the number and proportion of cells relative to parent populations and the full dataset (All) are shown here:
| Cell Type | Level(s) | N cells | % of Parent | % of All |
|---|---|---|---|---|
| B cell | 1 | 160,632 | NA | 8.82% |
| ├ Transitional B cell | 2, 3 | 12,555 | 7.82% | 0.69% |
| ├ Naive B cell | 2 | 86,711 | 53.98% | 4.76% |
| ┆ ├ Core naive B cell | 3 | 82,802 | 95.49% | 4.55% |
| ┆ └ ISG+ naive B cell | 3 | 3,909 | 4.51% | 0.22% |
| ├ Memory B cell | 2 | 47,886 | 29.81% | 2.63% |
| ┆ ├ Early memory B cell | 3 | 3,202 | 6.69% | 0.18% |
| ┆ ├ Core memory B cell | 3 | 39,718 | 82.94% | 2.18% |
| ┆ ├ Type 2 polarized memory B cell | 3 | 2,641 | 5.52% | 0.15% |
| ┆ ├ CD95 memory B cell | 3 | 1,892 | 3.95% | 0.10% |
| ┆ └ Activated memory B cell | 3 | 433 | 0.90% | 0.02% |
| ├ Effector B cell | 2 | 11,329 | 7.05% | 0.62% |
| ┆ ├ CD27+ effector B cell | 3 | 7,014 | 61.91% | 0.39% |
| ┆ ├ CD27- effector B cell | 3 | 4,315 | 38.09% | 0.24% |
| └ Plasma cell | 2, 3 | 2,151 | 1.34% | 0.12% |
Cell type definitions
Level 1
Here, we capture all peripheral blood populations with a B cell identity under a single label, spanning the full spectrum of B cell maturation. As such, the level 1 label captures a range of functional subsets from immature or transitional B cells, just emerging from the hematopoietic development process in bone marrow, to highly differentiated memory B cells and antibody-secreting or plasma cells, that arise after encountering their cognate antigen and undergoing further selection and maturation. We applied our knowledge of the distinct transcriptional and phenotypic features of human B cell populations to establish a subset classification based on single cell gene expression profiles.
In this reference we have one Level 1 label:
- B cell: We distinguished B cells, including plasma cells, from other peripheral immune cell populations based on expression of B cell receptor (BCR) complex genes. These include co-receptor genes CD19, CD79A, CD79B and CD21 as well as immunoglobulin genes, including the heavy chain (IgH) constant genes IGHM, IGHD, IGHA1, IGHA2, IGHG1, IGHG2, IGHG3, IGHG4, and IGHE and light chain constant genes IGKC,IGLC2, or IGLC3. MS4A1, which encodes the CD20 surface receptor, and PAX5 transcription factor gene expression have ubiquitous B cell expression, with Plasma cells as the exception. Furthermore, we confirmed positive expression of genes that enable antigen presentation, specifically class II major histocompatibility complex (MHC) loci such as HLA-DR and HLA-DQ. Plasma cells were expected to have slightly reduced class II MHC gene expression and significantly higher immunoglobulin gene expression levels.
Level 2
Here, we began to distinguish antigen-inexperienced B cell subsets, namely Transitional and Naive B cells, from antigen-experienced subsets, specifically Memory B cells and Plasma cells. Antigen-inexperienced B cells express only unswitched isotype functional BCR on their surface, and accordingly have higher IGHM and IGHD gene expression. Antigen-experienced B cell populations express either unswitched or switched isotype BCR on their surface and, as such, express high levels of both IGHM and IGHD (unswitched) and IGHG, IGHA, or IGHE (switched) class IgH genes. Plasma cells are highly enriched for class-switched isotype BCR and also have the highest levels of immunoglobulin gene expression among all B cell subsets, due to their function as antibody secreting cells.
At Level 2, we subdivided B cells into 5 major subclasses:
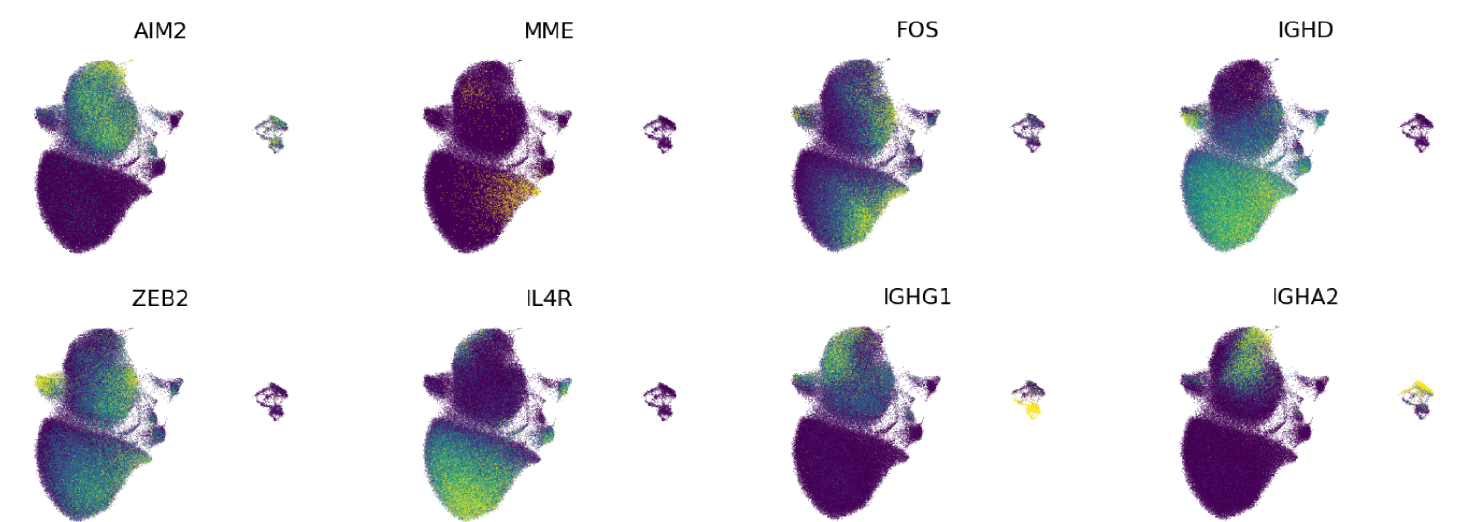
- Transitional B cell: Transitional or immature B cells are the population that first emerge into circulation after B cell development in the bone marrow. These cells can be distinguished from other subsets by higher MME, CD9, CD38, PAX5, and FCER2 expression.
- Naive B cell: Naive B cells are a more developed population than Transitional B cells. Their primary function is to circulate through blood and lymph between various lymphoid tissues in search of the cognate antigen of their unique BCR. This population expresses the IL4R and FCER2 genes similar to Transitional B cells, but have lower expression of MME, CD24, and CD9.
- Memory B cell: Memory B cells arise when Naive B cells encounter their cognate antigen and receive further activating signals, which support differentiation and affinity maturation (King, et al., 2021; Phad, et al., 2022; Glass, et al., 2020). This population is defined by CD27 and AIM2 gene expression. Memory cell populations also have higher expression of IgH constant region genes that specify ‘class-switched’ isotype, including IGHG1, IGHG2, IGHG3, IGHG4, IGHA1, IGHA2, and IGHE.
- Effector B cell: In this atlas, Effector B cells are a type of memory B cell with a distinct phenotype, defined by greater gene expression of ITGAX, FCRL4, FCRL5, TBX21, ZEB2, and PDCD1. These cells present a wide spectrum of CD27 and AIM2 expression, including non-expression. This memory population has been historically captured under a variety of names, including atypical B cells, CD27- IgD- or DN2 cells, CD21low B cells, FCRL4/5+ cells, Tbet+ B cells, CD11c+ CD19hi Effector B cells, and activated or age-associated memory B cells. The origins and function of this rare and distinctive memory population remains yet to be clearly elucidated in humans (Gao and Cockburn 2022). These cells arise in an antigen-responsive manner and are a component of the antigen-specific B cell population in the context of COVID and influenza vaccination (Sutton, et al., 2021; Glass, et al., 2020; Ellebedy et al., 2016; Priest et al., 2024). This population is also highly expanded in individuals with certain autoimmune conditions and those that have experienced multiple Malaria infections (Jenks, et al., 2018; Sutton et al., 2021).
- Plasma cell: This antibody-producing population is clearly distinguishable from the rest of the B cell compartment due to a large number of unique transcriptional features (Nutt et al. 2015; Phad et al. 2022; King et al. 2021). As mentioned earlier, this subset lacks expression of MS4A1 (also known as CD20) and PAX5, which are ubiquitously expressed by all other B cell populations. However, these cells sustain expression of another B cell lineage-defining receptor gene, CD19, when in the absence of immune perturbations such as vaccination or infections. Plasma cells also express subset-defining transcription factor and regulatory protein genes, namely PRDM1, XBP1, MZB1, and SLAMF7. Finally, these cells have especially high expression of the CD27 and CD38 genes as well as most of the BCR heavy chain constant region genes, excepting for less IGHD transcript.
Level 3
At Level 3 of our annotations, we further divided the Naive, Memory, and Effector B cells based on expression of specific gene programs. Transitional B cells and Plasma cells were not further subdivided.
Naive B cells were divided into 2 subpopulations:
- Core naive B cell: The same gene expression profile described for level 2 Naive B cells applies for this level 3 label. This population is the most abundant B cell subset in the periphery.
- ISG+ naive B cell: This small naive B cell subset is defined by a unique profile of interferon response-related gene expression. In this atlas, population-defining genes include STAT3, STAT1, IFI44L, and ISG15.
Memory B cells were divided into 5 subpopulations:
- Early memory B cell: The Early Memory B cell subset represents a transitional population between antigen-inexperienced and antigen-experienced B cells. Prior studies of this population’s surface phenotype and IgH complementarity-determining region 3 (CDR3) indicate that these are antigen-exposed cells with ‘intermediate’ memory phenotypes and CDR3 somatic mutational burdens (Glass, et al., 2020). Here, we define this population by intermediate expression of memory-defining genes, CD27, AIM2 and class-switched isotype IgH (IGHA1, IGHA2 and IGHG1, IGHG2, IGHG3, and IGHG4). ‘Intermediate’ is defined as being between level 2 Naive and Memory B cell median expression levels. It should be noted that this transcription-based labeling approach is less accurate than utilizing a surface marker-based approach, defined by the presence of CD45RB in a specific glycosylated form that can be detected with the MEM55 antibody clone (Koers, et al., 2022).
- Core memory B cell: This population represents the largest portion of the peripheral memory B cell compartment. The gene expression profile described for level 2 Memory B cells applies to this level 3 label without further gene expression requirements.
- Type 2 polarized memory B cell: In our atlas, we capture a recently defined, type 2 immunity-polarized memory population that is enriched for allergen-specific cells as well as IgE+ plasma cell precursors (Ota, et al., 2024; Koenig, et al., 2024) . These memory cells are distinguishable by expression of IL4R, FCER2, COCH, IGHG1, IGHG4, and IGHE. IGHE gene expression by memory B cells represents mostly germline transcription, given that it is primarily plasma cells that express functional IgE+ BCR or antibodies.
- CD95 memory B cell: CD95+ memory B cells are defined by high expression of the FAS gene, which encodes the CD95 protein, also called Fas cell surface death receptor (Glass, et al., 2020). This population also displays the highest AIM2 and class-switched isotype IgH (IGHA1, IGHA2, IGHG1, IGHG2, IGHG3, and IGHG4) gene expression among memory B cell subsets. The origins and functions of this memory subset in humans are not entirely clear, although it has been suggested that these cells could be recent germinal center emigrants.
- Activated memory B cell: This small memory B cell subset is defined by a unique profile of activation-related gene expression, including FOS, CD69, JUN, MCL1, and MYC. The detection and abundance of this population among healthy individuals varies greatly, and is likely a reflection that this label captures a distinguishable memory cell state rather than a stable, functional subset.
Effector B cells were split into 2 subpopulations:
- CD27+ effector B cell: This subset of Effector memory B cells is distinguished by positive expression of CD27 and tends to have more transcriptional overlap with other memory B cells than the CD27- population.
- CD27- effector B cell: This subset of Effector memory B cells is distinguished by a lack of CD27 expression. These cells also tend to have lower expression of AIM2 and higher expression of some subset-defining genes, such as ITGAX, TBX21, and ZEB2.
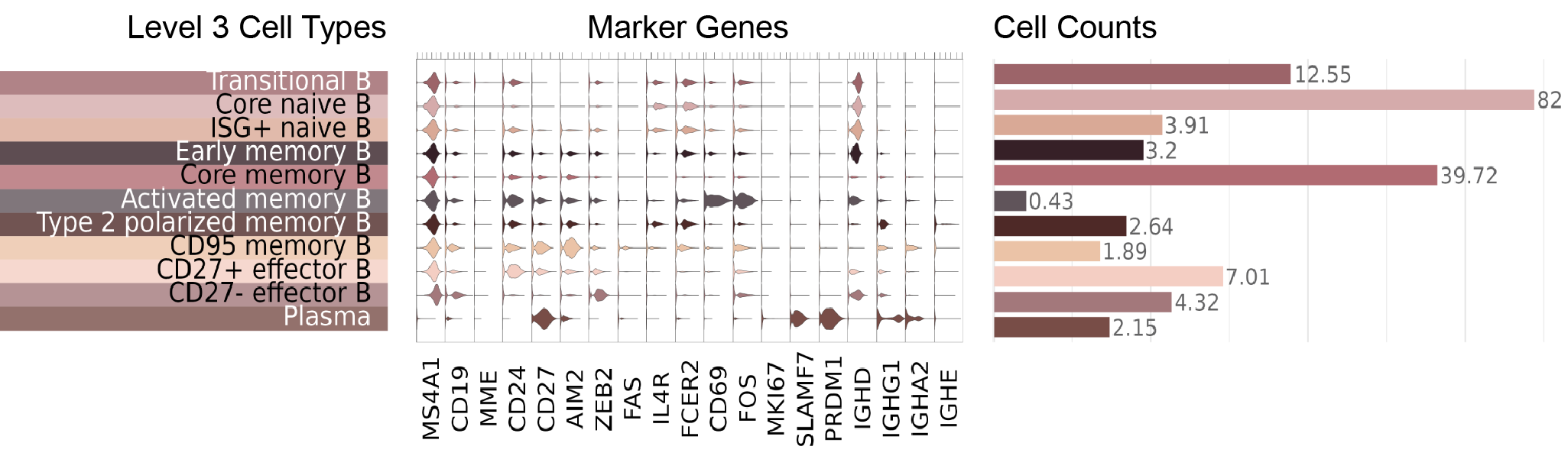
Marker visualizations
Key markers used to define NK and ILC cell types are shown in this figure, where we display a violin plot for each marker gene and each Level 3 cell type:
Expression of key marker genes shown on our UMAP projection:
References
Ellebedy AH, Jackson KJL, Kissick HT, Nakaya HI, Davis CW, Roskin KM, et al. Defining antigen-specific plasmablast and memory B cell subsets in human blood after viral infection or vaccination. Nat Immunol. 2016;17: 1226–1234.
doi:10.1038/ni.3533
Gao X, Cockburn IA. The development and function of CD11c+ atypical B cells - insights from single cell analysis. Front Immunol. 2022;13: 979060.
doi:10.3389/fimmu.2022.979060
Glass DR, Tsai AG, Oliveria JP, Hartmann FJ, Kimmey SC, Calderon AA, et al. An integrated multi-omic single-cell atlas of human B cell identity. Immunity. 2020;53: 217-232.e5.
doi:10.1016/j.immuni.2020.06.013
Jenks SA, Cashman KS, Zumaquero E, Marigorta UM, Patel AV, Wang X, et al. Distinct effector B cells induced by unregulated Toll-like receptor 7 contribute to pathogenic responses in systemic Lupus Erythematosus. Immunity. 2018;49: 725-739.e6.
doi:10.1016/j.immuni.2018.08.015
King HW, Orban N, Riches JC, Clear AJ, Warnes G, Teichmann SA, et al. Single-cell analysis of human B cell maturation predicts how antibody class switching shapes selection dynamics. Sci Immunol. 2021;6: eabe6291.
doi:10.1126/sciimmunol.abe6291
Koenig JFE, Knudsen NPH, Phelps A, Bruton K, Hoof I, Lund G, et al. Type 2-polarized memory B cells hold allergen-specific IgE memory. Sci Transl Med. 2024;16: eadi0944.
doi:10.1126/scitranslmed.adi0944
Koers J, Pollastro S, Tol S, Pico-Knijnenburg I, Derksen NIL, van Schouwenburg PA, et al. CD45RB glycosylation and Ig isotype define maturation of functionally distinct B cell subsets in human peripheral blood. Front Immunol. 2022;13: 891316.
doi:10.3389/fimmu.2022.891316
Nutt SL, Hodgkin PD, Tarlinton DM, Corcoran LM. The generation of antibody-secreting plasma cells. Nat Rev Immunol. 2015;15: 160–171.
doi:10.1038/nri3795
Ota M, Hoehn KB, Fernandes-Braga W, Ota T, Aranda CJ, Friedman S, et al. CD23+IgG1+ memory B cells are poised to switch to pathogenic IgE production in food allergy. Sci Transl Med. 2024;16: eadi0673.
doi:10.1126/scitranslmed.adi0673
Phad GE, Pinto D, Foglierini M, Akhmedov M, Rossi RL, Malvicini E, et al. Clonal structure, stability and dynamics of human memory B cells and circulating plasmablasts. Nat Immunol. 2022;23: 1076–1085.
doi:10.1038/s41590-022-01230-1
Priest DG, Ebihara T, Tulyeu J, Søndergaard JN, Sakakibara S, Sugihara F, et al. Atypical and non-classical CD45RBlo memory B cells are the majority of circulating SARS-CoV-2 specific B cells following mRNA vaccination or COVID-19. Nat Commun. 2024;15: 6811.
doi:10.1038/s41467-024-50997-4
Sutton HJ, Aye R, Idris AH, Vistein R, Nduati E, Kai O, et al. Atypical B cells are part of an alternative lineage of B cells that participates in responses to vaccination and infection in humans. Cell Rep. 2021;34: 108684.
doi:10.1016/j.celrep.2020.108684